Rhodochrosite
Rodocrosite
MnCO₃ Properties
- Category
- Mineral
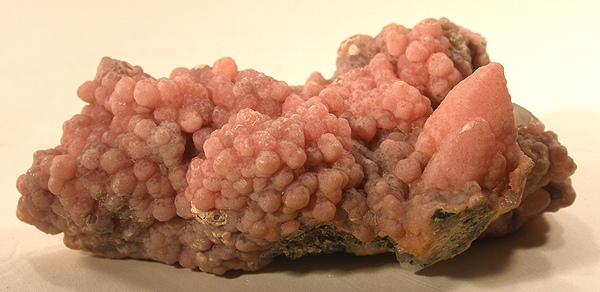
Rhodochrosite is a manganese carbonate (MnCO₃) that forms trigonal crystals of intense pink color, often striped with white, and is appreciated both as a collector mineral and as an ornamental gemstone.
Rhodochrosite is a *trigonal carbonate* of manganese that represents one of the most aesthetically fascinating minerals. It forms predominantly in low-temperature hydrothermal environments associated with manganese deposits, and sometimes in metamorphic rocks rich in manganese. Its characteristic pink color is due to the presence of Mn²⁺ ions, while the alternating white bands are often composed of calcite or variations in manganese concentration. Rhodochrosite was first discovered in Argentina (Catamarca), where the most spectacular specimens are still found, with clear crystals and perfectly defined striations. As a gemstone, it is relatively fragile (hardness 3.5–4 Mohs) and requires protection from wear; however, the finest examples command significant prices in the international gemmological market, especially those from historic Argentine or Peruvian deposits.
Rhodochrosite crystallizes in the *trigonal system*, space group R3c, with lattice parameters a = 4.777 Å and c = 15.67 Å. Density is approximately 3.5 g/cm³. The ordinary refractive index (nₒ) is ~1.597 and the extraordinary refractive index (nₑ) is ~1.816, with birefringence Δn ≈ 0.219. It exhibits *pleochroism* from nearly colorless to intense pink when observed in thin sections. Fluorescence under ultraviolet light (UV) is variable: some specimens show red fluorescence under long-wave UV (365 nm), while others remain inactive. The absorption spectrum presents characteristic bands around 540 nm and 630 nm due to d-d transitions of Mn²⁺. Rhodochrosite is soluble in dilute acids with effervescence. Quantitative chemical analysis typically reveals: MnO 61–62%, CO₂ 37–38%, with trace amounts of Ca, Fe, and Zn. Gemmological-quality specimens show absence of visible inclusions and saturated color; marked *pleochroism* and high birefringence are diagnostic features. Rhodochrosite frequently associates with *calcite*, *hematite*, *pyrite*, and *galena* in vein environments; in metamorphic contexts, it occurs with *spessartine* and *rhodonite*.
Mining localities
- Catamarca, Argentina (giacimento storico di Capillitas, cristalli rosa con striature bianche perfette)
- Perù — Ancash e Junín (esemplari di qualità gemmologica)
- Sud Africa — Transvaal (associate a depositi di manganese)
- Stati Uniti — Colorado e Montana (cristalli in filoni idrotermali)
- Cina — Hunan (rodocrosite massiva e cristallizzata)
- Romania — Banat (associata a mineralizzazioni di rame e manganese)
- Giappone — Iwate (cristalli di piccole dimensioni ma di buona qualità)
Frequently Asked Questions
How can you identify authentic rhodochrosite from other pink minerals?
Authentic rhodochrosite (MnCO₃) is recognized by its characteristic parallel white striations on intense pink crystals, hardness of 3.5-4 on the Mohs scale, and effervescence when exposed to dilute acids. Well-defined trigonal structures and uniform pink-magenta coloration are additional reliable indicators of authenticity.
What is the price of rhodochrosite and where can you buy it?
Rhodochrosite prices range from €10-50 for common specimens to €200-500+ for gem-quality crystals from Argentine Capillitas mines. It is available through specialized mineral shops, international gem fairs, and certified online mineral retailers.
Where is rhodochrosite found in the world?
Rhodochrosite forms primarily in secondary manganese deposits associated with manganese oxide ore bodies, with the finest crystals coming from Argentina (Capillitas, Catamarca), Romania, Peru, and Colorado in the USA. Argentina remains the world's leading producer of collectible and gem-quality specimens.
Why is rhodochrosite pink and what is its chemical composition?
Rhodochrosite is a pure manganese carbonate (MnCO₃) and its intense pink color derives from manganese ions (Mn²⁺) in the trigonal crystal structure, which selectively absorb certain light wavelengths. The white striations are caused by thin layers of impurities or slight compositional variations during crystallization.
Entry generated with Claude API (Anthropic) on data extracted from Mindat, RRUFF and Wikipedia. Not yet reviewed by a human expert. Verify data against original sources before citing in formal work.